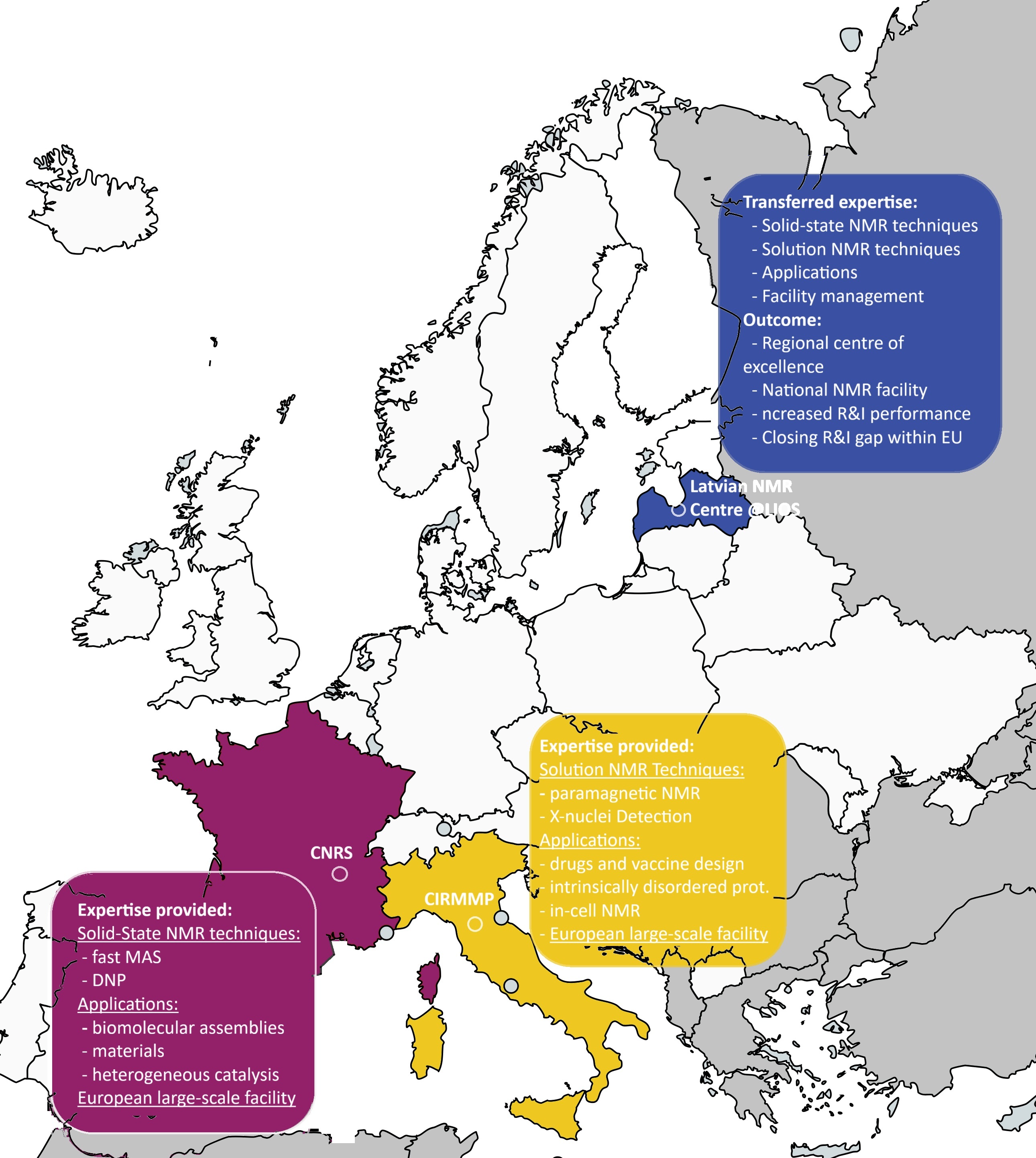
n just 17 months, MR LATVIA has moved from “project start” to real momentum: stronger skills at LIOS, deeper European collaboration, and a clear pathway to open access for researchers and innovators who need high-field NMR for (bio)pharma and beyond. Working side-by-side with two top European NMR centres - CNRS-CRMN (Lyon) and CERM/CIRMMP (Florence) - the consortium is building a practical, sustainable model for a Latvian NMR centre of excellence.
Why this matters for Latvia
MR LATVIA turns high-field NMR from a limited, specialist capability into a reliable national resource for both academia and industry. For universities and research institutes, it strengthens local expertise, accelerates advanced (bio)molecular research, and anchors Latvia more firmly in European collaboration networks. For companies - especially in (bio)pharma, biotech, materials and chemical development - it means faster, higher-confidence molecular characterisation and clearer routes to access expertise and instrumentation through a transparent core-facility ACCESS model, reducing the need to outsource abroad and supporting R&D competitiveness at home.
What has changed in 17 months?
People & skills (the fastest multiplier)
- 5 advanced scientific webinars delivered across partners.
- 19 staff exchange visits completed (LIOS ↔ CNRS-CRMN; LIOS ↔ CERM/CIRMMP), supporting hands-on method transfer and joint experimental work.
- Two flagship training actions completed: PANACEA WEEK-2025 in Lyon (May 2025) and the ITACA SB NMR Summer School + satellite exchange event in Florence (September 2025).
Early-stage researcher growth
- One two-month secondment completed; two secondments ongoing through mid-February 2026, embedding research-level NMR skills in the LIOS team.
Research progress: concrete milestones, real datasets
- Isotopically labelled proteins and peptide variants prepared and quality-confirmed by NMR
- VLP-coupled peptide variants prepared and quality-verified by NMR
- Spectral assignment for full-length proteins/peptide variants (monomeric and aggregated forms) initiated and ongoing.
- Survey of joint funding calls completed to support future collaborative proposals.
Opening the door: the ACCESS platform is ready
- MR LATVIA designed and approved the ACCESS platform, defining user categories, access rules, and pricing principles—validated with partner centres as test users.
- The first public ACCESS call is planned for February 2026, marking the shift from capacity-building to broader community impact.
Visibility & outreach
- Website and digital channels maintained; partner amplification across social media.
- 3 newsletters and 2 digital brochures produced for policy and industry stakeholders.
- Dissemination at key events including EUROMAR 2025 (Oulu), FEBS3+ Baltics (Vilnius), and the Balti Magnet Conference (Tallinn, Estonia).
What’s next
MR LATVIA now moves into a delivery-heavy phase: launching the public ACCESS call, continuing secondments and advanced training, and progressing spectral assignments toward complete datasets and joint outputs.
Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or the European Research Executive Agency. Neither the European Union nor the granting authority can be held responsible.

