On 26 November 2025, MR LATVIA hosted an online webinar dedicated to improving shared practices in NMR instrument scheduling and laboratory safety within the regional research community. The session was led by Rebecca Del Conte, Technical Manager at CERM/CIRMMP, who presented the operational approach used at CERM to manage access to high-field NMR infrastructure.
Del Conte outlined how NMR time requests—from internal researchers, EU project partners, and external users—are evaluated within a structured workflow that balances scientific needs, instrument constraints, maintenance schedules, and technician availability. A priority-based scheduling method ensures efficient allocation of machine time, beginning with cryogen refills and critical maintenance, followed by external users, specialised experiments, and all remaining requests. A coded scheduling system provides clear visibility of internal and external users, EU-funded time, maintenance windows, and instrument downtime.
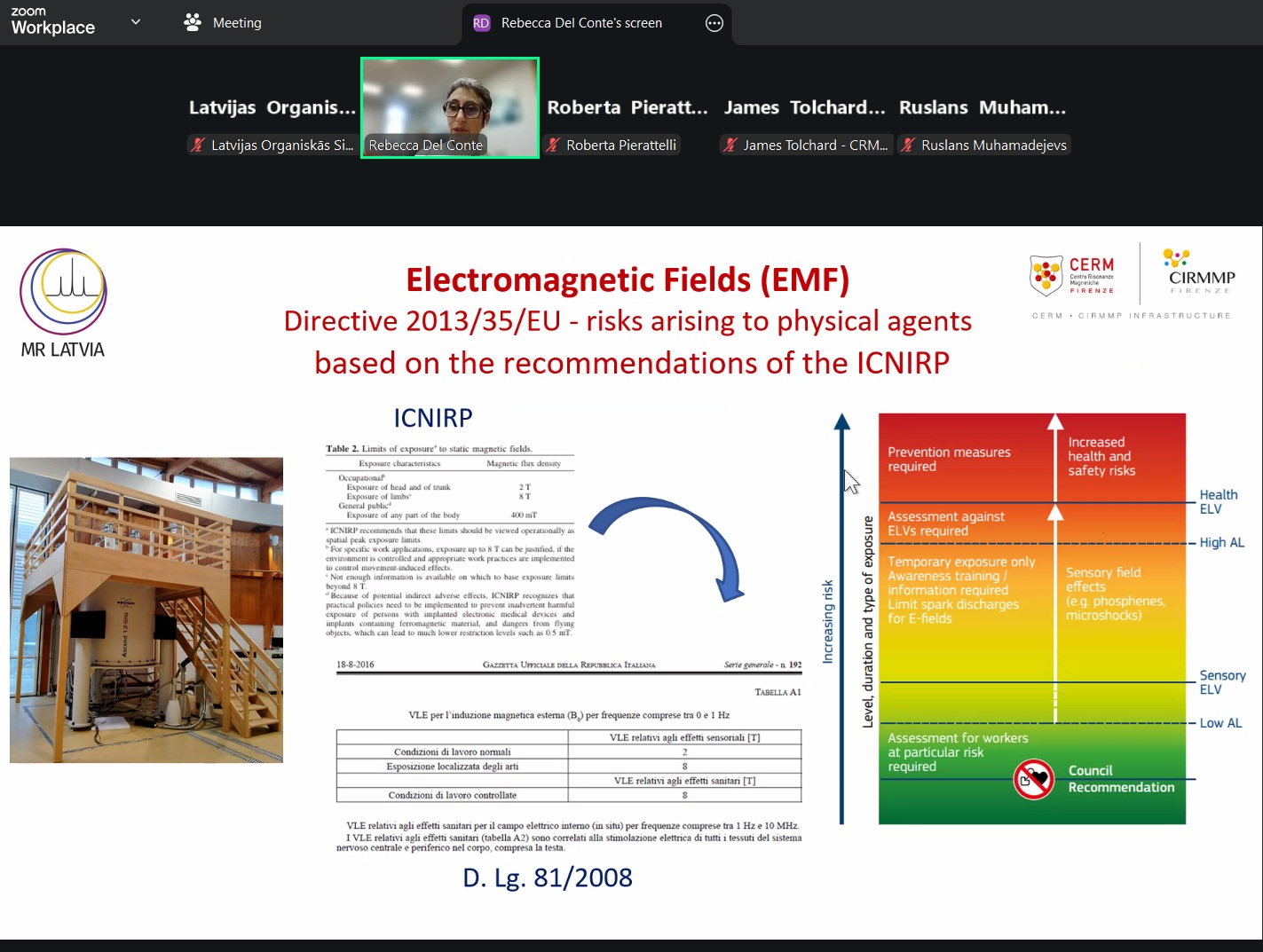
The webinar also highlighted CERM’s safety framework, which applies the ALARA principle and fully aligns with European and Italian regulations. Key risk areas—chemical agents, biological agents, cryogenic liquids, and electromagnetic fields generated by NMR magnets—were reviewed alongside the corresponding EU directives. Preventive measures include appropriate PPE, collective protection systems, oxygen monitoring, restricted EMF zones, and proper containment for biological materials. All users working in CERM laboratories must complete mandatory safety training and acknowledge the institution’s Safety and Risk Prevention Manual.
An open discussion enriched the session with perspectives from partner institutions. Representatives from CRMN shared their experience in managing high-demand NMR facilities and coordinating user access. From LIOS, Arcady Sobolev and Ruslans Muhamadejevs provided a detailed overview of safety procedures currently implemented at LIOS, including chemical and biological safety practices, cryogen handling protocols, EMF precautions, and user training requirements. Their contribution offered valuable insights into how safety systems are structured across different research environments.
The webinar demonstrated the importance of exchanging practical expertise across the MR LATVIA network and underscored the shared commitment to transparent scheduling, safe laboratory environments, and harmonised operational standards.

